There are different types of acid. The high concentration ones are of course the most dangerous.
Do not panic if you get a little 0.01 molar HCl on your finger, you silly duck!
Much of the power of an acid comes from the fact that it has a relationship with water.
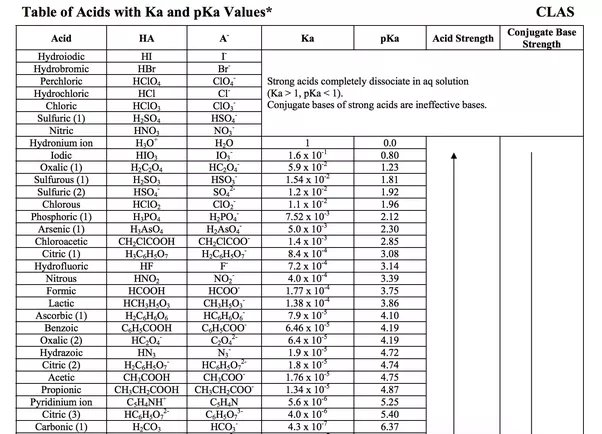
Table of Acids w Kas and pKas.pdf
Pure and dry acidic compounds are typically fairly boring things. In fact, hydrogen chloride is not even very soluble in water compared to its peers, and the strongest hydrochloric acid (HCL - Muriatic Acid).
Typically, though, when a strong acid begins to dissolve, it will release a tremendous amount of heat, which is why you are advised to never add water to a strong acid, since it is likely to cause the small amount of water initially added to boil, forming hot steam and aerosolized concentrated acid.
The act of adding acid to meat causes a strong dehydrating effect as well as increasing the temperature and turning water to steam. Water in the tissue is dissolved into the acid drying, heating, and cooking the tissues exposed.
In other words, acid as it is diluted by removing water from meat subsequently causes it to release substantial heat, causing significant thermal burns.
Acids, because they are often catalysts rather than actual reactants, can cause far more damage than say, poisons, which have critical doses to reach before they act as poisons.
INNER DAMAGE VS OUTER DAMAGE
INNER CELL:
- If acids get into the interior of a cell they lower the pH sufficiently to stop the cell biochemistry and they can denature (change the structure and function) of proteins such as enzymes.
- However the lipid (grease) layer of the cell wall prevents most aqueous material getting in.
- When irritant material such as acid gets into live tissue, the body tries to dilute it with water, giving swelling and blisters.
FLESH AND SKIN:
- Outside the cells are structural materials which in skin and flesh are mostly collagen, a protein, which means a polymer of amino acids.
- As the name suggests, amino acids have one end which is an amine (a base) and one end which is a carboxylic acid, and these have been reacted together (eliminating water) to form a link.
- You can reverse this process by acid-catalyzed hydrolysis, so aqueous acids can break down the structural material of your body. This is one of the main mechanisms of the damage caused by an acid burn.