Why chemical bonds are important to understand explosive reactions? Well, because they are the basis to understand the explosive compounds.
Where does the energy that is released when a chemical compound is exploded come from? The answer is actually on basic chemistry (and if you’ve read the title, you already know it), chemical bonds, more precisely on how they are formed and broken.
Here’s something that for me was not intuitive on the subject of how chemical bonds works:
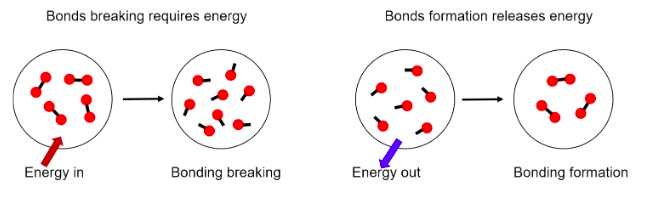
Now that’s something that you should remember, because explosives are compounds where some bonds will be formed to release a huge amount of energy. But why this energy is released?
Bonds are stable configurations of (relative) minimal energy. So when entering a bond the combined energy level of the contributing atoms is being reduced to a local minimum. That way “free energy” is released that shows as heat or radiation in the environment.
When a chemical reaction occurs, molecular bonds are broken and other bonds are formed to make different molecules. For example, the bonds of two water molecules are broken to form hydrogen and oxygen.
2H2O --> 2H2+O2When a chemical reaction occurs, the atoms in the reactants rearrange their chemical bonds to make products. The new arrangement of bonds does not have the same total energy as the bonds in the reactants. Therefore, when chemical reactions occur, there will always be an accompanying energy change.
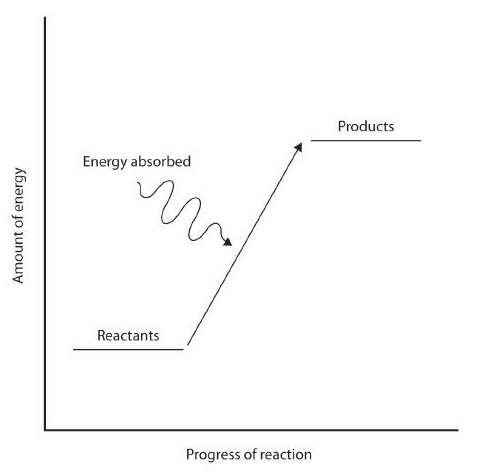
In some reactions, the energy of the products is lower than the energy of the reactants. Thus, in the course of the reaction, the substances lose energy to the surrounding environment. Such reactions are exothermic and can be represented by an energy-level diagram*.* In most cases, the energy is given off as heat (although a few reactions give off energy as light). In chemical reactions where the products have a higher energy than the reactants, the reactants must absorb energy from their environment to react. These reactions are endothermic and can be represented by an energy-level.
Yeah, a lot of funny reactions, and specially those that end with an explosions, are exothermic.
Energy is always required to break a bond, which is known as bond energy. When a bond is strong, there is a higher bond energy because it takes more energy to break a strong bond. This correlates with bond order and bond length. When the Bond order is higher, bond length is shorter, and the shorter the bond length means a greater the Bond Energy because of increased electric attraction. In general, the shorter the bond length, the greater the bond energy**.**
NITROGEN
Many common explosives are some carbon core covered in Nitrate groups, like TNT, PETN, and the most common explosive by weight is the Ammonium nitrate family (widely used in mining as it is cheap ammonium nitrate is fertilizer, and millions of tonnes are produced every year).
An explosive is a chemically unstable material, so it has energy that, with the right bump, will be released. Many (not all) explosives contain nitrogen compounds which have weak molecular bonds to other materials. Bump those bonds, they break, and reform to another material releasing energy as they do. That release of energy was the loud bang that you will heard on explosion.
The explosiveness of nitrogen compounds is driven by the huge release of energy that occurs when the nitrogen-nitrogen triple bonds form. One triple bond releases much more energy when it forms than is needed to break the three single bonds that bind the nitrogen in the starting compound.
“The point is that you are breaking weaker bonds and making a very strong bond”
A second factor makes nitrogen compounds explosive: the newly formed nitrogen molecules form a gas, which can expand very quickly and form a shock wave.
There are other compounds that are explosive that don’t contain nitrogen. The point about nitrogen explosives are that they are relatively stable and so don’t go off prematurely. So you can control them with triggers.
We can get nitrogen pretty stably attached to carbon and hydrogen by single bonds (ammonia and amines). Even organic imines (C=N double bonds) are pretty stable.
When you start attaching nitrogen to oxygen (nitroglycerine or trinitrotoluene or picric acid) or worse yet, to itself by single or double bonds (hydrazine or azobenzene or sodium azide or worst of all, diazomethane) you can very quickly reach the danger zone.
To anticipate a question “Why doesn’t nitric acid blow up?” the answer is, to the best of my knowledge, the fact that “concentrated” nitric acid is about 50% by weight (compare to industrial sulfuric acid, 98%). Diluting the stuff with water helps to keep it stable, and dilution also helps stabilize other explosive nitrogen compounds
But if you mix nitroglycerine with an inert solid like celite, you get dynamite; that stuff requires high heat or a shock from a detonator to go off.