Most chemical explosions involve a limited set of simple reactions, all of which involve oxidation (reaction with oxygen). A relatively easy way to balance chemical explosive equations is to assume that the following partial reactions take place to their maximum extent (meaning one of the reactants is totally consumed) and in order of precedence:
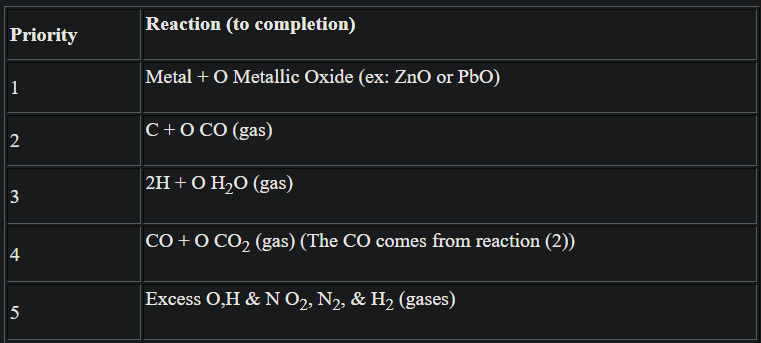
Example balance the combustion of TNT:
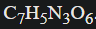
No metals, so start with priority 2:
6C + 60 6CO --> leaving 1C, 5H, 3N;No oxygen left, skip priorities 3 and 4. Lastly, the gases combine:
3N 3/2 N2
-------------
5H 5/2 H2, leaving 1 C not consumed.Overall:
C7-H5-N3-O6-6CO + 5/2 H2 + 3/2 N2 + C.The total amount of energy released ∆E (kJ/mol) in the reaction is called the heat of explosion. It can be calculated by comparing the heats of formation before and after the reaction (enthalpy):
∆E = ∆Ef(reactants) - ∆Ef(products)The heat of explosion is defined so that it will be positive for an exothermic reaction.
heats of formation for the products and many common explosives (reactants):
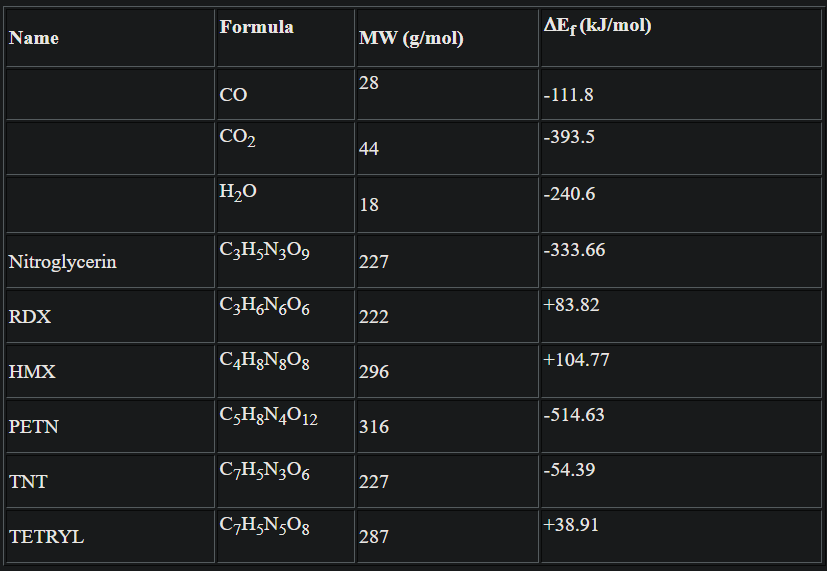
Example: find the heat of explosion for TNT.
Before (TNT):
∆Ef = -54.4 kJ/mol
After (Reaction):
∆Ef = 6(-111.8) + 5/2(0) + 3/2(0) + 1(0) = -670.8 kJ/mol
∆E = (-54.4) + 670.8 - = 616.4 kJ/mol,
Since DE > 0, the reaction is exothermic, and the heat of explosion is +616.4 kJ/mol.
Expressed on a mass basis, TNT releases (kJ/mol):
(1000 J/1 kJ)(1 mol/227 g) = 2175 J/g.
--> 1 kg of TNT releases 2.175 x 10^6 J of energy.Since most of the energy release comes from oxidation reactions, the amount of oxygen available is a critical factor. If there is insufficient oxygen to react with the available carbon and hydrogen, the explosive is considered to be oxygen deficient. The converse is considered oxygen rich. A quantitative measure of this is called the oxygen balance:
C,H,M&O are the number of moles of carbon, hydrogen, metal and oxygen in the balanced reaction and MW is the molecular weight of oxygen (= 16 g/mol) or the explosive.
Find the oxygen balance for TNT:
OB = -(100 %)(16/227)[2(7) + 5/2 - 6] = -72%
- As a general rule, the oxygen balance should be near zero to get the maximum amount of energy release. Other concerns like stability or volatility often limit the oxygen balance for chemical compounds. TNT is an example of a relatively powerful explosive that is oxygen deficient.