The determining factor in the conversion of the heat of explosion into mechanical work is the amount of product gases available for expansion. In the case of TNT, 10 moles of gas are produced for each mole of explosive. We can exploit this fact to make predictions about the actual explosive strength of other chemicals. This is known as the Berthelot approximation, which states that the relative explosive strength of a material (as compared to TNT on a mass basis) may be calculated on the basis of two factors:
- The change in internal energy (∆E) and
- The amount of gas produced.
If we combine these factors and put in values for our reference, TNT, we obtain:
Relative Strength (%) = 840 ∆n * ∆E / (MW^2)
- ∆n = the number of moles of gas per mole of explosive.
- ∆E = the heat of explosion in kJ/mol
- MW = molecular weight of explosive in g/mol
- The factor of 840 accounts for the units and values of DE and Dn for TNT.
Calculate the Berthelot relative strength for RDX:
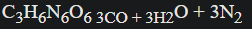
- MW = 222 g/mol
- ∆n = 9 mol
∆Ef (before) = 83.82 kJ/mol
∆Ef (after) = 3(-111.8) + 3(-240.6) = -1057.2 kJ/mol
RS = 840 (9) (83.82 + 1057.2)/(222^2)
RS = 175 %
The relative explosive strength calculated in this manner is of limited use. What is really important is the actual strength which can only be measured by experiment.