Thermite is a composition of a metal powder and a metal oxide. When ignited (requires considerable amount of energy) it undergoes an exothermic reduction-oxidation reaction, that releases a lot of energy.
Always, when we want to burn something, we need a fuel and a oxidizer (like an car engine, you need fuel, but you also need air inside combustion chamber), with thermite it’s not different.
There’s no such fixed thermite composition recipe, but some elements are common:
-
FUELS
-
OXIDIZERS
Here’s the classical thermite recipe:
The reactants (Iron and Aluminum in this case) are commonly powdered and mixed with a binder to keep the material solid and prevent separation.
This composition requires a really high activation energy which means it’s very hard to light on fire. So besides that, you might want include some starter mix.
IGNITION
REDACTED
This content has been redacted by the Department of Truth™. The information previously contained in this document has been deemed inappropriate for public distribution.
THEORY
So let’s see what is happening behind the scenes…
The thermite reaction occurs between a metal and a metal oxide, where the metal is higher on the reactivity series than the one in the oxide. The metals on the top of the table are more reactive than the ones listed to the bottom.
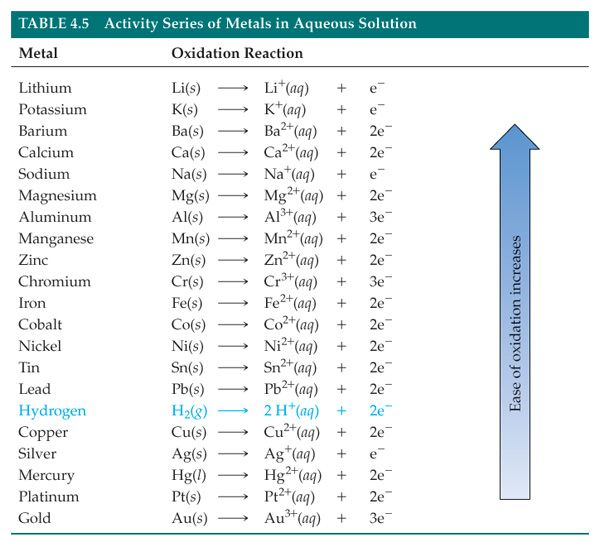
You can see that, Aluminum is above iron, and the blue arrow say that the element that is higher, is more easy to suffer oxidation. So we can say that Aluminum can reduce the oxide of iron oxide.
Note that sometimes the reaction will not happen, because even if you pick the right reactive elements, the activation energy is too high!
The reaction can be described as:
Fe2O3 + 2 Al --> 2 Fe + Al2O3 ΔHº = -850 kJAluminum reduces the oxide of iron oxide, in other words, aluminum replaces the iron in the oxide, because aluminum forms stronger and more stable bonds with oxygen than iron. The products are aluminum oxide, elemental iron and heat… a lot of heat.
MORE DESTRUCTIVE
In general, an increase in the volume of gaseous reaction products of a thermite blend increases the heat transfer rate (and therefore damage) of that particular thermite blend.
It is usually used with other ingredients that increase its incendiary effects.
Thermate-TH3:
Similar to the classical thermite, thermate consists of a mixture of very fine aluminium metal powder and iron(III) oxide, but also has in its composition elemental sulfur and barium nitrate, which serve to boost the thermite reaction.
The variant of the Thermate, called Thermate-TH3, is a mixture of thermite and pyrotechnic additives that have been found superior to standard thermite for incendiary purposes. Its composition by weight is generally:
- 68.7% - THERMITE
- 29.0% - BARIUM NITRATE
- 2.00% - SULFUR
- 0.30% - BINDER (such as PBAN).
The addition of barium nitrate to thermite increases it’s thermal effect, produces a larger flame, and significantly reduces the ignition temperature. Although the primary purpose of Thermate-TH3 by the armed forces is as an incendiary anti-materiel weapon, it also has uses in welding together metal components.
Warning: Thermate is more unstable than thermite!
Copper (II) oxide (Black):
3CuO(s) + 2Al(s) → Al2O3(s) + 3Cu(l); ΔH = -1203.8 kJ/molThis thermite mixture have an incredibly fast burn rate. Additionally copper has a comparatively low melting point, and this mix should be treated as extremely dangerous. Due to the amount of molten copper produced, and the mixtures tendency to explode (caused by the high burn rate), this mixture is known for sending out thousands of drops of liquid metal, and it is recommended that you be very careful when attempting to ignite this compound.
According to the reaction’s stoichiometry, the ratio of Copper (II) Oxide to Aluminum powder by weight is about 4.4:1, or more exactly, 4.42:1.
Copper (I) oxide (Red):
This one are dangerous like the previous one!
3Cu2O(s) + 2Al(s) → Al2O3(s) + 6Cu(l); ΔH = -1169.8 kJ/molAccording to the reaction’s stoichiometry, the ratio of Copper (I) oxide to Aluminum powder by weight is about 8:1, or more exactly, 7.96:1.
Manganese (IV) oxide (Black):
3MnO2(s) + 4Al(s) → 2Al2O3(s) + 3Mn(l); ΔH = -1788.7 kJ/molAccording to the reaction’s stoichiometry, the ratio of Manganese (IV) oxide to Aluminum powder by weight is about 2.4:1, or more exactly, 2.42:1.
Iron (II,III) oxide (Black):
Fe2O3(s) + 2Al(s) → Al2O3(s) + 2Fe(l); ΔH = -851.5 kJ/mol3Fe3O4(s) + 8Al(s) → 4Al2O3(s) + 9Fe(l); ΔH = -3347.6 kJ/molThough the ΔH for this reaction as balanced is higher than that for Fe2O3 thermite, a given mass of Fe2O3 thermite will generate more energy than the same mass of Fe3O4 thermite. According to the reaction’s stoichiometry, the ratio of Iron (II,III) oxide to Aluminum powder by weight is about 3.2:1, or more exactly, 3.22:1.