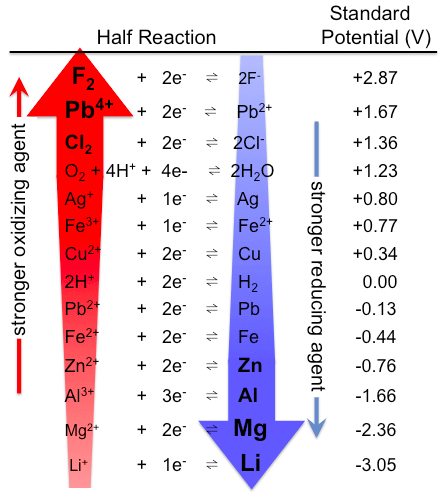
An oxidation-reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. An oxidation-reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron. Redox reactions are common and vital to some of the basic functions of life, including photosynthesis, respiration, combustion, and corrosion or rusting.
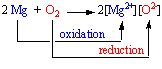
Because electrons are neither created nor destroyed in a chemical reaction, oxidation and reduction are linked. It is impossible to have one without the other, as shown in the figure below.
The Activity Series: Predicting Spontaneous Redox Reactions
Single-replacement reactions only occur when the element that is doing the replacing is more reactive than the element that is being replaced. Therefore, it is useful to have a list of elements in order of their relative reactivities. The activity series is a list of elements in decreasing order of their reactivity.
Since metals replace other metals, while nonmetals replace other nonmetals, they each have a separate activity series. The table below is an activity series of most common metals and of the halogens.
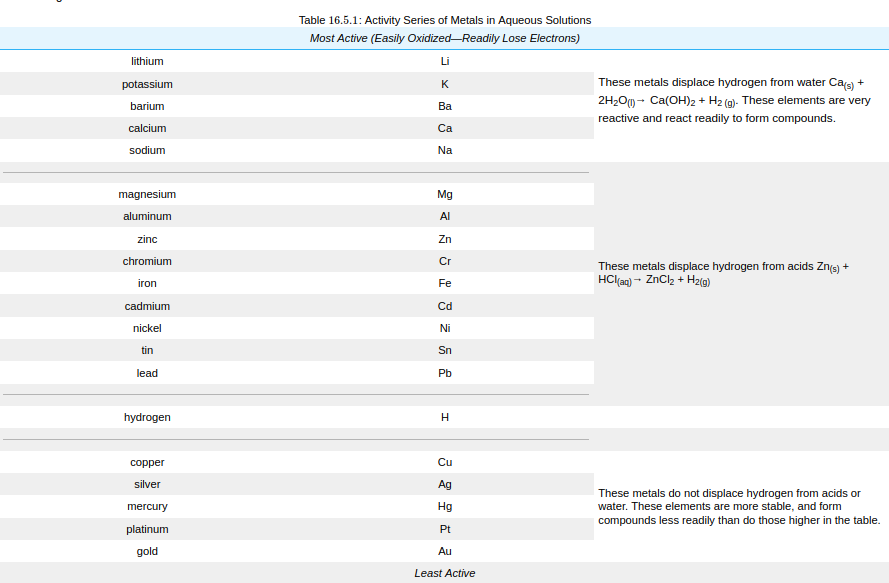
For a single-replacement reaction, a given element is capable of replacing an element that is below it in the activity series. This can be used to predict if a reaction will occur. Suppose that small pieces of the metal nickel were placed into two separate aqueous solutions: one of iron (III) nitrate and one of lead (II) nitrate. Looking at the activity series, we see that nickel is below iron, but above lead. Therefore, the nickel metal will be capable of replacing the lead in a reaction, but will not be capable of replacing iron.
Ni(s) + Pb(NO3)2(aq) --> Ni(NO3)2(aq) + Pb(s)Ni(s) + Fe(NO3)3(aq) --> NR (no reaction)In the descriptions that accompany the activity series of metals, a given metal is also capable of undergoing the reactions. For example, lithium will react with cold water, replacing hydrogen. It will also react with steam and with acids, since that requires a lower degree of reactivity.