CHEMICAL BURN THOUGHTS
First things first, here the word “burn” has two meanings…
- Damage to skin and other tissue due to high temperature. This **thermal **burning breaks down the chemical nature of the tissue.
- Damage to the skin due to contact with a “corrosive” chemical. Again tissue is broken down this time by a chemical burn.
The first thing to understand about chemical burns, is that it usually happens when some acid or base get in contact with the body. People usually assumes that only acids are dangerous, but bases are very effective and powerful too (sometimes even more aggressive than acids).
If you’ve watched “Fight Club” you probably remember about the remarkable chemical burn scene (that is a metaphor for some philosophy shitty, that is not important here).

In this scene, the characters are making soap, which uses NaOH (Sodium Hydroxide) on recipe. Tyler uses NaOH, a powerful and aggressive base, to provoke the chemical burn. Later, Tyler uses CH3COOH (vinegar), an acid, to neutralize the reaction, stopping the chemical burn.
CH3COOH(aq) + NaOH(aq) --> CH3COONa(aq) + H2OThat’s a good example of a dangerous base in action. Do not underestimate the power of the bases or the acids, both of then can cause a lot of pain.
(Rubber is your friend here, use rubber wear)
Acid strength (strong acid/weak acid) does not necessarily determine how dangerous that acid is!
Some acids hydrolize the surface of your skin. Basically it really messes up the proteins and lipids on your skin. So acids “burn” not mainly through thermal means but by basically breaking your proteins and lipids through chemical reactions. There can be a thermal component on acid burns. Some acids cause chemical reactions with skin that lead to dehydration of organic molecules found on your skin. This rapid dehydration is exothermic and can cause thermal damage.
This is why Muriatic Acid (HCl) is not as damaging as Sulfuric Acid (H2SO4) even though HCl is usually the stronger acid.
Another example of a very dangerous acid is Hydrofluoric Acid (HF). Hydrofluoric acid is a weak acid but it is probably one of the nastiest acids out there. In its vapor form it will penetrate your skin and the fluoride ions can bind to the calcium found in your bones to form CaF. Basically it takes those Ca ions needed for nerves and makes it into insoluble CaF…you will die because of a heart attack. Acids can be nasty or can be as harmless as acetic acid (all depending on molarity of course). I hope I could answer your question but I’m not an expert so I’m sure a better answer will come along
REACTION BETWEEN LOVERS
Acid/Base reactions are pretty straight forward, it’s a middle school, basic chemistry subject.
I always like to think about it as a classical romance cliche, where the opposites match somehow. On the chemical world, this match happens between to powerful - sometimes dangerous - opposite chemical compounds and produces something neutral (?).
Here is the general rule to understand the basic reaction between an acid and a base: it produces salt and water (as show on image… I wrote it in the case of selective blindness)
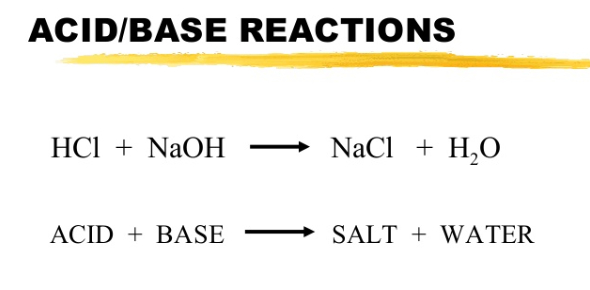
What you should know:
- Acid formulas start with an H, forms H+ ions
- Base formulas end with an OH, forms OH- ions
- Both ions (H+ and OH-) will dissociate in water
Some examples:
- HNO3 is nitric acid, starts with H
- HCl is Hydrochloric **acid, **tarts with H
- H2CO3 is carbonic acid, starts with H
- NH4OH is a base - ammonium hydroxide and ends with OH
- KOH is a base - Potassium hydroxide and ends with OH
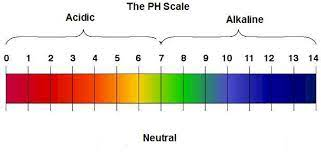
The pH
pH measure how strong a base or an acid aqueous is
There’s not much to say about pH. If the pH is less than 7, you have an acid solution. If the pH is greater than 7, you have a alkaline (base) solution.
https://ch302.cm.utexas.edu/chemEQ/neutralizations/selector.php?name=identify-acid-base-salt